Nitrogen Loss as Ammonia from Unincorporated Surface-applied UAN and Urea
Research was funded by the Tennessee Department of Agriculture (Land and Water Stewardship Programs)
Nutifafa Adotey, Assistant Professor and Soil and Nutrient Specialist, Samuel Okai, Graduate Students, Xinhua (Frank) Yin, Professor & Cropping System Scientist, Daniel Yoder, Professor, Lori Duncan, Assistant Professor and Row Crop Sustainability Specialist, Debasish Saha, Assistant Professor and Soil Scientist
Introduction
In Tennessee, the primary N source used are urea and urea ammonium nitrate (UAN). Other sources of N fertilizers such as anhydrous ammonia is used in some counties in Central and West Tennessee. It is a known fact that ammonia volatilization from applied N fertilizers may occurs from (1) unincorporated, surface-applied urea-based fertilizer; (2) anhydrous ammonia due to poor equipment calibration; and (3) unincorporated, surface-applied ammonium-based fertilizer on alkaline soils. This N loss from these fertilizers is govern by a complex interaction of prevailing environmental conditions, soil properties and management practices. Soil properties determine the potential of ammonia loss while the extent of the loss is dictated by environmental conditions and management practices. Urea hydrolysis is limited in UAN; therefore, no ammonia is formed from the urea portion of UAN compared to urea which undergoes complete hydrolysis. Generally, ammonia volatilization from surface application of UAN tends to be lower than surface application of urea. Some studies, however, found higher ammonia loss from UAN than urea in alkaline soils. A study that demonstrates the amount of N loss as ammonia across different soil types will encourage producers to pay close attention to their N management strategies and adopt efficient N management practices.
Materials and Method
A laboratory study was carried out to evaluate ammonia volatilization from urea and UAN applied onto the surface of 12 soils collected from producer fields. The experiment was evaluated using a precise in-lab environment-controlled system (picture below). To initiate the volatility study, the glass chambers were filled with 500 g of air‐dried soil and moistened to two‐thirds field capacity of each soil. The moistened soil is then incubated at a constant temperature of 26°C for 48 h prior to fertilizer application. At the end of incubation, N fertilizer was surface applied at an equivalent rate of 120 lbs. N a–1 based on the exposed soil surface area within the glass chambers. Ammonia from N fertilizer and soil were captured in 100 mL of 0.02 M orthophosphoric acid on 1, 2, 3, 4, 5, 6, 7, 9, 11, 13, and 16 days after fertilizer application.

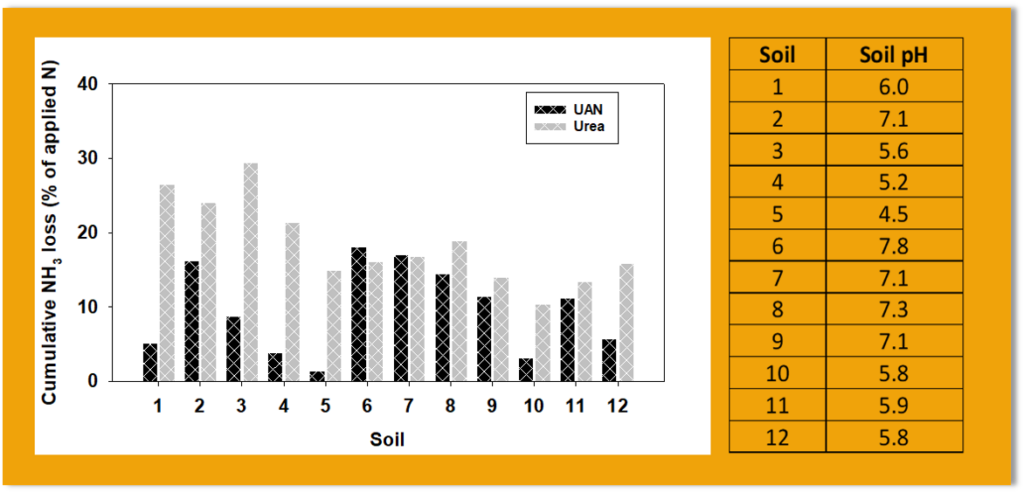
Results and Discussion
Cumulative ammonia loss from urea ranged from 10 – 29% of applied N and UAN ranged from 1 – 18% of applied N. Cumulative ammonia loss from urea was significantly higher than those from UAN in soils 1, 2, 3, 4, 5, 10 and 12 (Figure 1), while differences were not observed in the remaining soils. The composition of the N fertilizers and the initial soil pH may partly explain this observation. In regards to composition, urea ammonium nitrate (UAN) is made up of urea, ammonium, and nitrate, in contrast to urea-N fertilizer which is composed of 100 % urea. Applied urea undergoes urea hydrolysis to produce ammonium and bicarbonate, which results in an increase soil pH around the immediate environment of urea granule. In poorly buffered soils, the increase in soil pH may substantially drive ammonia volatilization from urea. However, in well buffered soil, the increase in pH may not drive ammonia loss from urea. Ammonia loss from applied UAN is not dependent on urea hydrolysis but the initial soil pH. In alkaline soils, the high soil pH favors the conversion of ammonium to ammonia so the initial concentration of ammonium in UAN results in higher ammonia loss than urea.
In this experiment, the pH of soil 1, 2, 3, 4, 5, 10, and 12 were less than 7 except for soil 2 meaning we don’t expect substantial ammonia loss from UAN. However, the loss of ammonia from urea could be higher or low depending on the buffering capacity of the soil. In high pH soils as seen in soil 2, 6, 7, 8, and 9, ammonia loss from urea was relatively higher than the low pH soils and, in most instances, similar to urea on corresponding soils. The initial pH plays a major role in ammonia loss from surface applied UAN.
Field studies in Georgia have reported that UAN lost a similar or higher amount of ammonia than urea, which were attributed to the influence of critical relative humidity (CRH). The low CRH (18 – 20 %) of UAN than urea (72 – 80 %) meant that UAN absorbed moisture from its surrounding more rapidly than urea, which would only absorb moisture when its relative humidity is around 80 %.
Tennessee producers have multiple options to successfully minimize ammonia loss when applying UAN in field with pH greater than 7. Surface-applied UAN should be treated with a proven nitrogen stabilizer for optimal yields when soils pH is above 7. Subsurface-banding and injection have been reported to reduce ammonia loss from UAN applied in fields with pH less than 7; however, there is limited information on the effect of these placement methods on ammonia loss when soil pH is greater than 7. Information on N stabilizers evaluated under growing conditions are available at https://utia.tennessee.edu/publications/wp-content/uploads/sites/269/2024/03/W1221.pdf and https://utia.tennessee.edu/publications/wp-content/uploads/sites/269/2023/10/PB1888.pdf.
Further reading
Adotey, N., McClure, A.T., Yin, X., and J. McNeal. 2024. Performance of enhanced efficiency nitrogen fertilizers and sidedress nitrogen placement methods in dryland corn. UT Extension Publication, W1221.
Adotey, N., McClure, A.T., Yin, X., and J. McNeal. 2021. Enhanced efficiency nitrogen fertilizer as a tool to control nitrogen loss in row crop production. UT Extension Publication, PB 1888
Cabrera, M. L., Franklin, D., Kissel, D. E., & Rema, J. (2023) Ammonia Volatilization from Urea and Urea-Ammonium Nitrate Solution Applied to a Grassland in the Southeastern USA. [Abstract]. ASA, CSSA, SSSA International Annual Meeting, St. Louis, MO. https://scisoc.confex.com/scisoc/2023am/meetingapp.cgi/Paper/153164
Vaio N., Cabrera M.L., Kissel D., Rema J.A., Newsome J.F., Calvert V.H. (2008) Ammonia volatilization from urea‐based fertilizers applied to tall fescue pastures in Georgia, USA. Soil Science Society of America Journal 72:1665-1671. DOI: 10.2136/sssaj2007.0300.





